What Is the Chemical Machinery That Neurons Use to Communicate
Explicate the mode neurons communicate
All functions performed by the nervous system—from a unproblematic motor reflex to more advanced functions similar making a memory or a decision—require neurons to communicate with one some other. While humans use words and body language to communicate, neurons use electrical and chemical signals. Just like a person in a committee, 1 neuron usually receives and synthesizes messages from multiple other neurons before "making the decision" to ship the message on to other neurons.
Learning Objectives
- Describe the basis of the resting membrane potential
- Explain the stages of an action potential and how action potentials are propagated
- Explain the similarities and differences betwixt chemical and electrical synapses
- Describe the process of signal summation
- Discuss the processes involved in synaptic plasticity
Resting Membrane Potential
For the nervous system to office, neurons must be able to send and receive signals. These signals are possible considering each neuron has a charged cellular membrane (a voltage difference between the within and the outside), and the charge of this membrane tin modify in response to neurotransmitter molecules released from other neurons and environmental stimuli. To understand how neurons communicate, one must first understand the basis of the baseline or "resting" membrane charge.
Neuronal Charged Membranes
The lipid bilayer membrane that surrounds a neuron is impermeable to charged molecules or ions. To enter or go out the neuron, ions must pass through special proteins called ion channels that span the membrane. Ion channels have dissimilar configurations: open, airtight, and inactive, as illustrated in Effigy 1. Some ion channels need to be activated in order to open up and let ions to laissez passer into or out of the cell. These ion channels are sensitive to the environment and can modify their shape accordingly. Ion channels that alter their construction in response to voltage changes are chosen voltage-gated ion channels. Voltage-gated ion channels regulate the relative concentrations of different ions within and outside the prison cell. The departure in full charge between the inside and outside of the jail cell is chosen the membrane potential.

Effigy i. Voltage-gated ion channels open in response to changes in membrane voltage. After activation, they become inactivated for a brief period and will no longer open up in response to a signal.
This video discusses the basis of the resting membrane potential.
Resting Membrane Potential
A neuron at rest is negatively charged: the inside of a cell is approximately 70 millivolts more negative than the outside (−70 mV, note that this number varies past neuron blazon and by species). This voltage is called the resting membrane potential; it is caused by differences in the concentrations of ions inside and exterior the cell. If the membrane were every bit permeable to all ions, each type of ion would flow across the membrane and the arrangement would accomplish equilibrium. Because ions cannot simply cross the membrane at will, there are different concentrations of several ions inside and outside the prison cell, every bit shown in Table 1.
| Tabular array 1. Ion Concentration Inside and Outside Neurons | |||
|---|---|---|---|
| Ion | Extracellular concentration (mM) | Intracellular concentration (mM) | Ratio exterior/inside |
| Na+ | 145 | 12 | 12 |
| Grand+ | 4 | 155 | 0.026 |
| Cl− | 120 | 4 | 30 |
| Organic anions (A−) | — | 100 | |
The resting membrane potential is a event of unlike concentrations inside and outside the cell. The divergence in the number of positively charged potassium ions (K+) within and outside the cell dominates the resting membrane potential (Effigy 2).

Figure 2. The (a) resting membrane potential is a result of dissimilar concentrations of Na+ and 1000+ ions inside and outside the prison cell. A nerve impulse causes Na+ to enter the cell, resulting in (b) depolarization. At the superlative action potential, K+ channels open up and the cell becomes (c) hyperpolarized.
When the membrane is at residue, M+ ions accumulate within the cell due to a net movement with the concentration gradient. The negative resting membrane potential is created and maintained by increasing the concentration of cations outside the jail cell (in the extracellular fluid) relative to inside the prison cell (in the cytoplasm). The negative accuse within the cell is created by the cell membrane being more permeable to potassium ion movement than sodium ion movement. In neurons, potassium ions are maintained at high concentrations inside the cell while sodium ions are maintained at high concentrations outside of the prison cell. The cell possesses potassium and sodium leakage channels that allow the two cations to diffuse downwards their concentration gradient.
Even so, the neurons have far more than potassium leakage channels than sodium leakage channels. Therefore, potassium diffuses out of the cell at a much faster rate than sodium leaks in. Considering more than cations are leaving the cell than are entering, this causes the interior of the prison cell to be negatively charged relative to the exterior of the cell. The actions of the sodium potassium pump assistance to maintain the resting potential, one time established. Call up that sodium potassium pumps brings two K+ ions into the jail cell while removing three Na+ ions per ATP consumed. Equally more cations are expelled from the prison cell than taken in, the inside of the cell remains negatively charged relative to the extracellular fluid. It should be noted that calcium ions (Cl–) tend to accrue outside of the prison cell because they are repelled by negatively-charged proteins within the cytoplasm.
Action Potential
A neuron can receive input from other neurons and, if this input is strong enough, send the betoken to downstream neurons. Transmission of a signal betwixt neurons is generally carried past a chemical chosen a neurotransmitter. Transmission of a bespeak inside a neuron (from dendrite to axon terminal) is carried by a brief reversal of the resting membrane potential chosen an activeness potential. When neurotransmitter molecules bind to receptors located on a neuron's dendrites, ion channels open. At excitatory synapses, this opening allows positive ions to enter the neuron and results in depolarization of the membrane—a decrease in the difference in voltage betwixt the within and outside of the neuron. A stimulus from a sensory cell or some other neuron depolarizes the target neuron to its threshold potential (−55 mV). Na+ channels in the axon hillock open up, allowing positive ions to enter the cell (Figure iii).
Once the sodium channels open, the neuron completely depolarizes to a membrane potential of nearly +xl mV. Action potentials are considered an "all-or nothing" upshot, in that, once the threshold potential is reached, the neuron always completely depolarizes. Once depolarization is consummate, the cell must now "reset" its membrane voltage back to the resting potential. To accomplish this, the Na+channels close and cannot be opened. This begins the neuron's refractory flow, in which it cannot produce another activity potential because its sodium channels volition not open up. At the aforementioned time, voltage-gated K+ channels open, allowing K+ to leave the cell. As K+ ions leave the cell, the membrane potential once again becomes negative. The diffusion of M+ out of the cell actually hyperpolarizes the jail cell, in that the membrane potential becomes more negative than the cell's normal resting potential. At this bespeak, the sodium channels will render to their resting state, pregnant they are ready to open again if the membrane potential once more exceeds the threshold potential. Somewhen the extra K+ ions diffuse out of the cell through the potassium leakage channels, bringing the cell from its hyperpolarized state, dorsum to its resting membrane potential.
Practice Question
The germination of an activity potential can be divided into five steps, which tin can be seen in Figure 3.
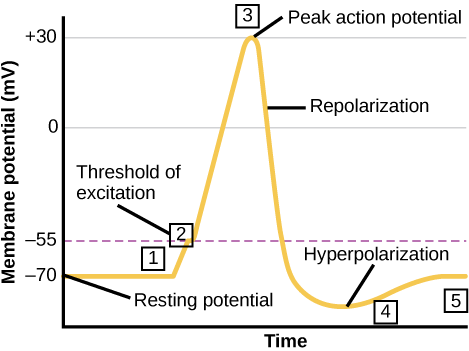
Figure 3. Activity Potential
- A stimulus from a sensory cell or another neuron causes the target cell to depolarize toward the threshold potential.
- If the threshold of excitation is reached, all Na+ channels open and the membrane depolarizes.
- At the tiptop action potential, Yard+ channels open and K+ begins to leave the cell. At the same time, Na+ channels close.
- The membrane becomes hyperpolarized every bit K+ ions continue to leave the cell. The hyperpolarized membrane is in a refractory period and cannot burn down.
- The Thou+ channels close and the Na+/Chiliad+ transporter restores the resting potential.
Potassium channel blockers, such as amiodarone and procainamide, which are used to care for abnormal electrical action in the heart, called cardiac dysrhythmia, impede the movement of Yard+ through voltage-gated K+ channels. Which office of the action potential would you wait potassium channels to bear on?
Show Answer
Potassium channel blockers slow the repolarization stage, but have no result on depolarization.
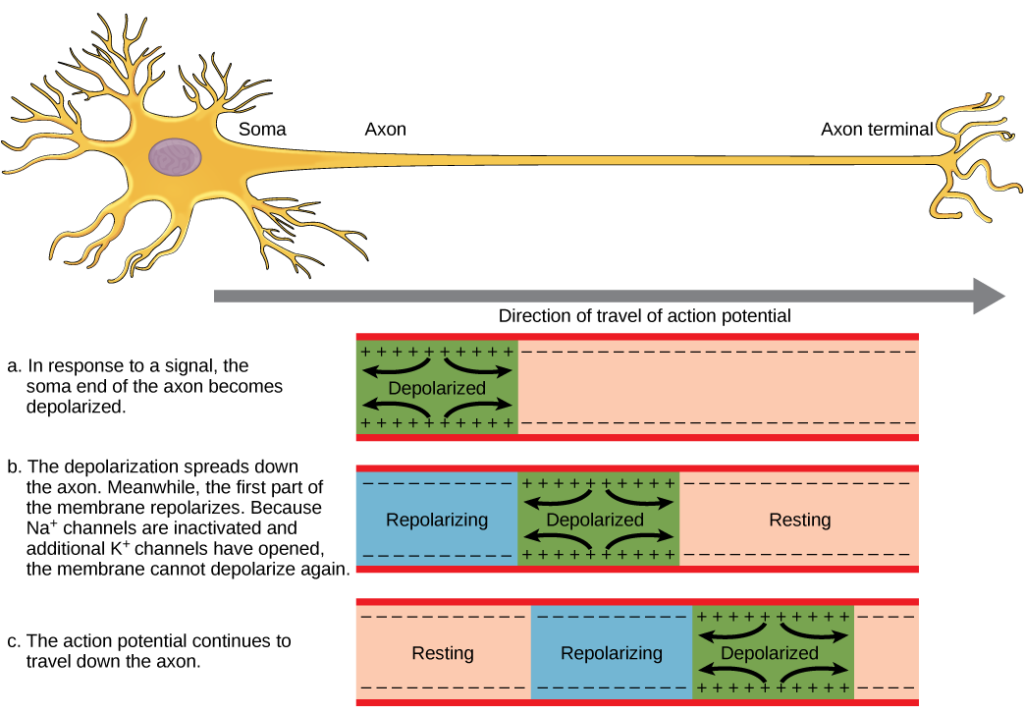
Figure four. The activity potential is conducted downwardly the axon as the axon membrane depolarizes, and so repolarizes.
This video presents an overview of activeness potential.
Myelin and the Propagation of the Action Potential
For an activeness potential to communicate information to another neuron, it must travel along the axon and reach the axon terminals where it tin can initiate neurotransmitter release. The speed of conduction of an action potential forth an axon is influenced by both the diameter of the axon and the axon'south resistance to current leak. Myelin acts as an insulator that prevents current from leaving the axon; this increases the speed of action potential conduction. In demyelinating diseases similar multiple sclerosis, action potential conduction slows considering electric current leaks from previously insulated axon areas.
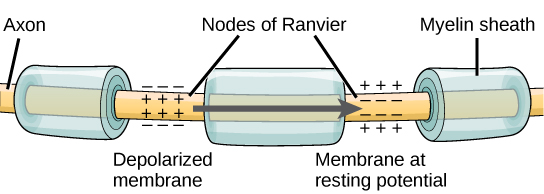
Effigy 5. Nodes of Ranvier are gaps in myelin coverage forth axons. Nodes comprise voltage-gated One thousand+ and Na+ channels. Action potentials travel down the axon by jumping from one node to the next.
The nodes of Ranvier, illustrated in Effigy 5 are gaps in the myelin sheath along the axon. These unmyelinated spaces are about one micrometer long and contain voltage gated Na+ and K+ channels. Menses of ions through these channels, especially the Na+ channels, regenerates the action potential over and over once more forth the axon. This 'jumping' of the action potential from ane node to the next is called saltatory conduction. If nodes of Ranvier were not present along an axon, the action potential would propagate very slowly since Na+ and K+ channels would have to continuously regenerate action potentials at every indicate forth the axon instead of at specific points. Nodes of Ranvier also save energy for the neuron since the channels only need to be nowadays at the nodes and not along the unabridged axon.
Chemic and Electrical Synapses
The synapse or "gap" is the place where information is transmitted from i neuron to another. Synapses usually class between axon terminals and dendritic spines, but this is non universally true. There are also axon-to-axon, dendrite-to-dendrite, and axon-to-jail cell trunk synapses. The neuron transmitting the signal is called the presynaptic neuron, and the neuron receiving the betoken is called the postsynaptic neuron. Notation that these designations are relative to a particular synapse—most neurons are both presynaptic and postsynaptic. In that location are two types of synapses: chemic and electrical.
Chemic Synapse

Effigy 6. This pseudocolored image taken with a scanning electron microscope shows an axon terminal that was broken open to reveal synaptic vesicles (blue and orange) inside the neuron. (credit: modification of work by Tina Carvalho, NIH-NIGMS; scale-bar information from Matt Russell)
When an action potential reaches the axon terminal information technology depolarizes the membrane and opens voltage-gated Na+ channels. Na+ ions enter the cell, farther depolarizing the presynaptic membrane. This depolarization causes voltage-gated Ca2+ channels to open. Calcium ions entering the cell initiate a signaling cascade that causes pocket-sized membrane-jump vesicles, chosen synaptic vesicles, containing neurotransmitter molecules to fuse with the presynaptic membrane. Synaptic vesicles are shown in Figure 6, which is an paradigm from a scanning electron microscope.
Fusion of a vesicle with the presynaptic membrane causes neurotransmitter to exist released into the synaptic cleft, the extracellular space betwixt the presynaptic and postsynaptic membranes, as illustrated in Figure 7. The neurotransmitter diffuses across the synaptic cleft and binds to receptor proteins on the postsynaptic membrane.

Figure 7. Communication at chemical synapses requires release of neurotransmitters. When the presynaptic membrane is depolarized, voltage-gated Ca2+ channels open and allow Ca2+ to enter the cell. The calcium entry causes synaptic vesicles to fuse with the membrane and release neurotransmitter molecules into the synaptic cleft. The neurotransmitter diffuses beyond the synaptic cleft and binds to ligand-gated ion channels in the postsynaptic membrane, resulting in a localized depolarization or hyperpolarization of the postsynaptic neuron.
The binding of a specific neurotransmitter causes item ion channels, in this case ligand-gated channels, on the postsynaptic membrane to open up. Neurotransmitters tin either accept excitatory or inhibitory effects on the postsynaptic membrane. There are several examples of well known neurotransmitters detailed in Table 1. For example, when acetylcholine is released at the synapse between a nervus and muscle (called the neuromuscular junction) by a presynaptic neuron, it causes postsynaptic Na+ channels to open up. Na+ enters the postsynaptic cell and causes the postsynaptic membrane to depolarize. This depolarization is called an excitatory postsynaptic potential (EPSP) and makes the postsynaptic neuron more than probable to burn an action potential. Release of neurotransmitter at inhibitory synapses causes inhibitory postsynaptic potentials (IPSPs), a hyperpolarization of the presynaptic membrane. For example, when the neurotransmitter GABA (gamma-aminobutyric acid) is released from a presynaptic neuron, it binds to and opens Cl– channels. Cl– ions enter the prison cell and hyperpolarizes the membrane, making the neuron less likely to fire an action potential.
In one case neurotransmission has occurred, the neurotransmitter must be removed from the synaptic cleft so the postsynaptic membrane tin can "reset" and be gear up to receive some other signal. This can be achieved in three ways: the neurotransmitter can diffuse abroad from the synaptic cleft, it can be degraded by enzymes in the synaptic cleft, or it can exist recycled (sometimes chosen reuptake) by the presynaptic neuron. Several drugs human action at this footstep of neurotransmission. For example, some drugs that are given to Alzheimer'due south patients work by inhibiting acetylcholinesterase, the enzyme that degrades acetylcholine. This inhibition of the enzyme substantially increases neurotransmission at synapses that release acetylcholine. Once released, the acetylcholine stays in the cleft and can continually demark and unbind to postsynaptic receptors.
| Table one. Neurotransmitters | ||
|---|---|---|
| Neurotransmitter | Function | Location |
| Acetylcholine | muscle control, memory | CNS and/or PNS |
| Serotonin | intestinal movement, mood regulation, slumber | gut, CNS |
| Dopamine | voluntary musculus movements, cognition, reward pathways | hypothalamus |
| Norepinephrine | fight or flight response | adrenal medulla |
| GABA | inhibits CNS | brain |
| Glutamate | by and large an excitatory neurotransmitter, memory | CNS, PNS |
Electrical Synapse
While electric synapses are fewer in number than chemical synapses, they are establish in all nervous systems and play important and unique roles. The fashion of neurotransmission in electrical synapses is quite unlike from that in chemic synapses. In an electrical synapse, the presynaptic and postsynaptic membranes are very close together and are really physically connected past channel proteins forming gap junctions. Gap junctions allow electric current to pass directly from one prison cell to the next. In addition to the ions that conduct this electric current, other molecules, such as ATP, can lengthened through the large gap junction pores.
At that place are key differences between chemical and electrical synapses. Considering chemic synapses depend on the release of neurotransmitter molecules from synaptic vesicles to pass on their signal, there is an approximately one millisecond delay betwixt when the axon potential reaches the presynaptic terminal and when the neurotransmitter leads to opening of postsynaptic ion channels. Additionally, this signaling is unidirectional. Signaling in electrical synapses, in contrast, is almost instantaneous (which is important for synapses involved in fundamental reflexes), and some electrical synapses are bidirectional. Electrical synapses are also more reliable as they are less likely to be blocked, and they are important for synchronizing the electrical activity of a group of neurons. For example, electrical synapses in the thalamus are idea to regulate slow-wave sleep, and disruption of these synapses can cause seizures.
Signal Summation
Sometimes a unmarried EPSP is strong plenty to induce an activeness potential in the postsynaptic neuron, simply ofttimes multiple presynaptic inputs must create EPSPs around the same fourth dimension for the postsynaptic neuron to be sufficiently depolarized to fire an activity potential. This procedure is called summation and occurs at the axon hillock, every bit illustrated in Figure 8. Additionally, one neuron frequently has inputs from many presynaptic neurons—some excitatory and some inhibitory—so IPSPs can cancel out EPSPs and vice versa. Information technology is the internet change in postsynaptic membrane voltage that determines whether the postsynaptic cell has reached its threshold of excitation needed to fire an action potential. Together, synaptic summation and the threshold for excitation human action as a filter so that random "noise" in the organization is not transmitted as important information.

Figure eight. A single neuron can receive both excitatory and inhibitory inputs from multiple neurons, resulting in local membrane depolarization (EPSP input) and hyperpolarization (IPSP input). All these inputs are added together at the axon hillock. If the EPSPs are strong enough to overcome the IPSPs and reach the threshold of excitation, the neuron volition burn down.
Brain-figurer interface
Amyotrophic lateral sclerosis (ALS, also called Lou Gehrig'south Disease) is a neurological disease characterized by the degeneration of the motor neurons that command voluntary movements. The disease begins with muscle weakening and lack of coordination and eventually destroys the neurons that control speech, breathing, and swallowing; in the cease, the disease can lead to paralysis. At that signal, patients crave assistance from machines to be able to breathe and to communicate. Several special technologies take been developed to allow "locked-in" patients to communicate with the residue of the world. Ane technology, for example, allows patients to blazon out sentences past twitching their cheek. These sentences can so exist read aloud by a computer.
A relatively new line of enquiry for helping paralyzed patients, including those with ALS, to communicate and retain a caste of cocky-sufficiency is called brain-estimator interface (BCI) engineering science and is illustrated in Figure 9. This technology sounds similar something out of science fiction: information technology allows paralyzed patients to control a computer using only their thoughts. At that place are several forms of BCI. Some forms use EEG recordings from electrodes taped onto the skull. These recordings comprise information from large populations of neurons that can be decoded past a computer. Other forms of BCI require the implantation of an array of electrodes smaller than a postage postage stamp in the arm and hand area of the motor cortex. This form of BCI, while more than invasive, is very powerful as each electrode can record actual action potentials from one or more neurons. These signals are so sent to a computer, which has been trained to decode the bespeak and feed it to a tool—such as a cursor on a calculator screen. This means that a patient with ALS tin use e-mail, read the Cyberspace, and communicate with others past thinking of moving his or her paw or arm (even though the paralyzed patient cannot make that bodily movement). Recent advances have immune a paralyzed locked-in patient who suffered a stroke xv years ago to control a robotic arm and fifty-fifty to feed herself coffee using BCI engineering.
Despite the amazing advancements in BCI engineering science, information technology also has limitations. The engineering tin can require many hours of training and long periods of intense concentration for the patient; it tin besides crave brain surgery to implant the devices.

Effigy 9. With brain-computer interface technology, neural signals from a paralyzed patient are collected, decoded, and and then fed to a tool, such as a computer, a wheelchair, or a robotic arm.
Lookout this video in which a paralyzed woman employ a encephalon-controlled robotic arm to bring a drink to her oral cavity, amid other images of brain-computer interface technology in activity.
Synaptic Plasticity
Synapses are non static structures. They can be weakened or strengthened. They can be cleaved, and new synapses can exist made. Synaptic plasticity allows for these changes, which are all needed for a operation nervous organisation. In fact, synaptic plasticity is the ground of learning and memory. Two processes in item, long-term potentiation (LTP) and long-term depression (LTD) are important forms of synaptic plasticity that occur in synapses in the hippocampus, a brain region that is involved in storing memories.
Long-term Potentiation (LTP)
Long-term potentiation (LTP) is a persistent strengthening of a synaptic connexion. LTP is based on the Hebbian principle: cells that burn together wire together. There are diverse mechanisms, none fully understood, behind the synaptic strengthening seen with LTP. I known machinery involves a type of postsynaptic glutamate receptor, called NMDA (Due north-Methyl-D-aspartate) receptors, shown in Figure x.

Figure 10. Calcium entry through postsynaptic NMDA receptors can initiate two different forms of synaptic plasticity: long-term potentiation (LTP) and long-term depression (LTD). LTP arises when a unmarried synapse is repeatedly stimulated. This stimulation causes a calcium- and CaMKII-dependent cellular cascade, which results in the insertion of more AMPA receptors into the postsynaptic membrane. The next time glutamate is released from the presynaptic cell, it will bind to both NMDA and the newly inserted AMPA receptors, thus depolarizing the membrane more than efficiently. LTD occurs when few glutamate molecules bind to NMDA receptors at a synapse (due to a depression firing rate of the presynaptic neuron). The calcium that does flow through NMDA receptors initiates a unlike calcineurin and protein phosphatase 1-dependent pour, which results in the endocytosis of AMPA receptors. This makes the postsynaptic neuron less responsive to glutamate released from the presynaptic neuron.
These receptors are commonly blocked by magnesium ions; nevertheless, when the postsynaptic neuron is depolarized by multiple presynaptic inputs in quick succession (either from one neuron or multiple neurons), the magnesium ions are forced out assuasive Ca ions to pass into the postsynaptic cell. Side by side, Caii+ ions entering the jail cell initiate a signaling cascade that causes a different type of glutamate receptor, called AMPA (α-amino-3-hydroxy-5-methyl-four-isoxazolepropionic acid) receptors, to exist inserted into the postsynaptic membrane, since activated AMPA receptors allow positive ions to enter the cell. So, the next time glutamate is released from the presynaptic membrane, it will have a larger excitatory upshot (EPSP) on the postsynaptic cell because the bounden of glutamate to these AMPA receptors will permit more positive ions into the cell. The insertion of additional AMPA receptors strengthens the synapse and ways that the postsynaptic neuron is more likely to burn in response to presynaptic neurotransmitter release. Some drugs of abuse co-opt the LTP pathway, and this synaptic strengthening can atomic number 82 to addiction.
Long-term Low (LTD)
Long-term depression (LTD) is essentially the contrary of LTP: it is a long-term weakening of a synaptic connectedness. 1 mechanism known to cause LTD too involves AMPA receptors. In this situation, calcium that enters through NMDA receptors initiates a dissimilar signaling cascade, which results in the removal of AMPA receptors from the postsynaptic membrane, as illustrated in Figure 10. The decrease in AMPA receptors in the membrane makes the postsynaptic neuron less responsive to glutamate released from the presynaptic neuron. While information technology may seem counterintuitive, LTD may be just every bit important for learning and memory as LTP. The weakening and pruning of unused synapses allows for unimportant connections to be lost and makes the synapses that have undergone LTP that much stronger past comparing.
Check Your Understanding
Respond the question(s) below to see how well yous understand the topics covered in the previous department. This curt quiz doesnot count toward your grade in the class, and y'all tin retake it an unlimited number of times.
Use this quiz to check your understanding and make up one's mind whether to (1) written report the previous department further or (ii) move on to the side by side section.
Source: https://courses.lumenlearning.com/wmopen-biology2/chapter/neuron-communication/
0 Response to "What Is the Chemical Machinery That Neurons Use to Communicate"
Post a Comment